|
5/24/2023 0 Comments Andexxa indication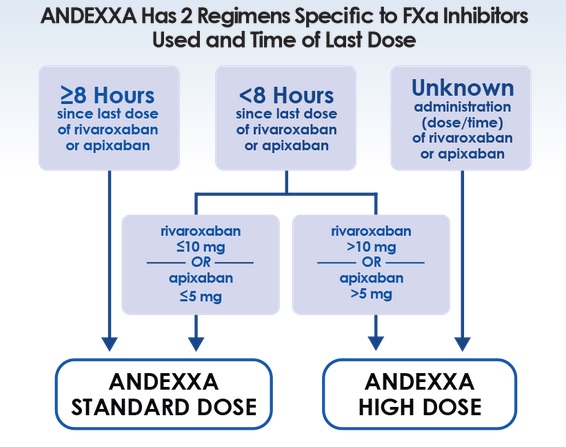
This data element matches the “Document Type” field of the Structured Product Listing.ĪCTIVE PRODUCT INCLUDED in the NDC Directory Indicates the type of product, such as Human Prescription Drug or Human Over the Counter Drug. Product Type What kind of product is this? Each active ingredient name is the preferred term of the UNII code submitted. Substance Name What is the Substance Name?Īn active ingredient is the substance responsible for the medicinal effects of a product specified by the substance's molecular structure or if the molecular structure is not known, defined by an unambiguous definition that identifies the substance. The generic name usually includes the active ingredient(s) of the product. The non-proprietary name is sometimes called the generic name. Non-Proprietary Name What is the Non-Proprietary Name? 
The proprietary name also known as the trade name is the name of the product chosen by the medication labeler for marketing purposes. Proprietary Name What is the Proprietary Name?
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
